- P.A. PBP3 INHIBITION DRIVERS
- P.A. PBP3 INHIBITION SKIN
- P.A. PBP3 INHIBITION TRIAL
- P.A. PBP3 INHIBITION PLUS
Sulbactam is a semi-synthetic penicillanic acid that was among the first β-lactamase inhibitors developed, in combination with ampicillin, for the treatment of infections caused by β-lactamase-producing bacterial pathogens ( Adnan et al., 2013). Sulbactam: A β-Lactamase Inhibitor With Intrinsic Antibacterial Activity Against Acinetobacter The key features of this unusual, dual BLI combination therapy are described below.
P.A. PBP3 INHIBITION PLUS
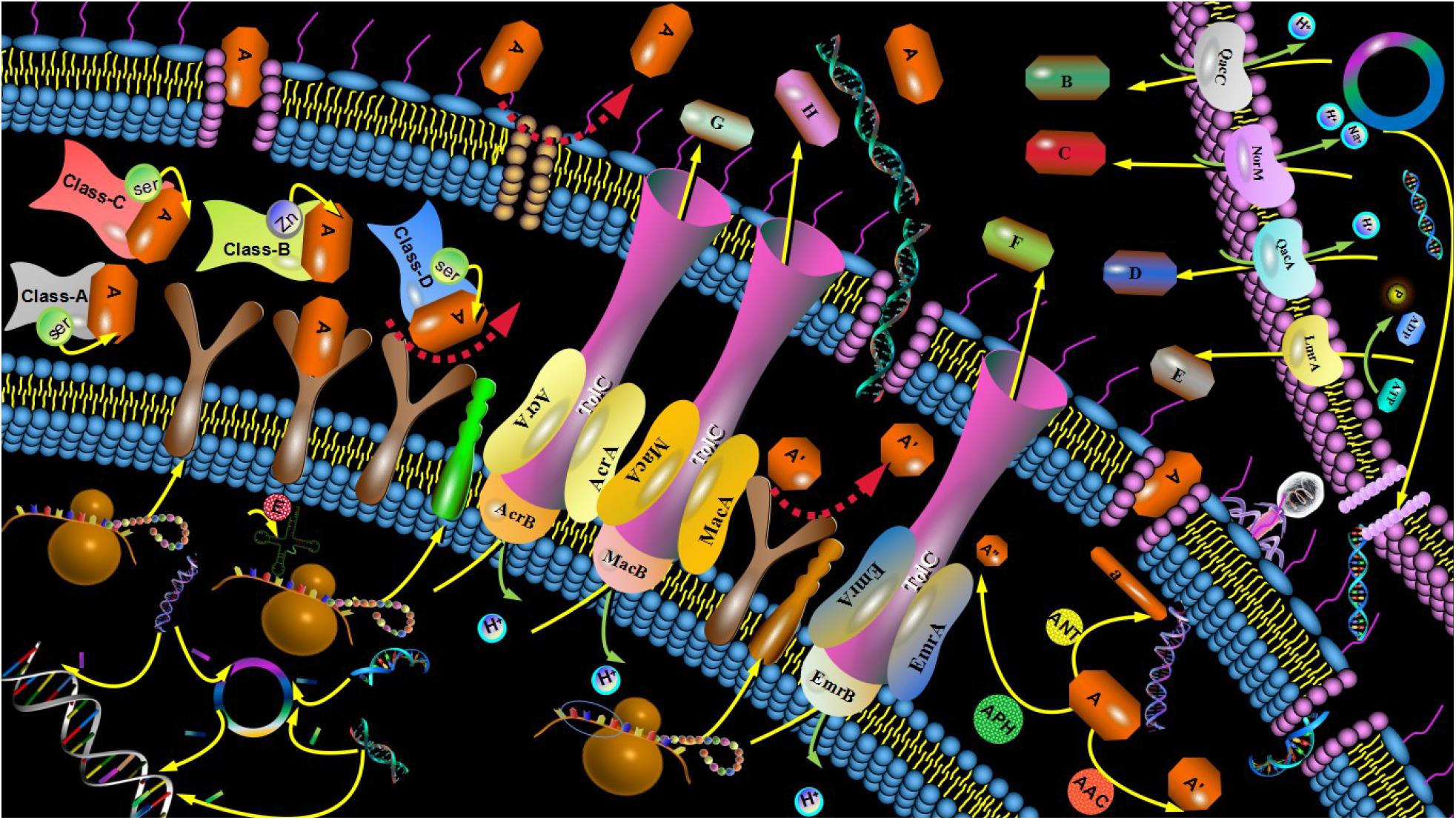
The only agent currently in late-stage clinical development for this indication is a combination of sulbactam, a first generation β-lactamase inhibitor (BLI) with intrinsic antibacterial activity against Acinetobacter spp., plus durlobactam, a next generation diazabicyclooctane (DBO) β-lactamase inhibitor with broad-spectrum activity against Class A, C, and D β-lactamases ( Durand-Reville et al., 2017). These findings have recently been proposed to be related to liabilities associated with siderophore-mediated uptake leading to heteroresistance ( Choby et al., 2021).
P.A. PBP3 INHIBITION TRIAL
baumannii bloodstream infections or nosocomial pneumonia in the recent CREDIBLE-CR Phase 3 trial ( Bassetti et al., 2021).

In addition, treatment with this agent resulted in higher mortality rates as compared to best available therapy in patients with A.

Although cefiderocol (Fetroja ®), which was recently approved for the treatment of drug-resistant Gram-negative pathogens, has potent in vitro activity against MDR Acinetobacter ( Isler et al., 2019), its in vivo efficacy in preclinical infection models of infection against cefiderocol-susceptible Acinetobacter baumannii is quite variable ( Monogue et al., 2017). In the past few decades, resistance to carbapenems in Acinetobacter and even the last resort agent colistin, has also increased at alarming rates worldwide ( Lee et al., 2017).ĭespite the significant unmet medical need, there are currently no reliably effective antibiotics for the treatment of carbapenem-resistant Acinetobacter infections. Clinical resistance in this organism to nearly all antibiotic classes, including cephalosporins, fluoroquinolones, aminoglycosides, and tetracyclines, is widespread and continues to increase ( Magiorakos et al., 2012 Wong et al., 2017). The incidence of these infections varies widely across the globe, ranging from 1% of surgical site infections and 12% of VAP in the United States ( Weiner et al., 2016) to 35% of all hospital-acquired drug-resistant infections in China ( Zhang et al., 2019). Most Acinetobacter infections are chronic, with mortality rates of 40–60% ( Wong et al., 2017).
P.A. PBP3 INHIBITION SKIN
These pathogens cause hospital-acquired or ventilator-associated pneumonia (VAP), bacteremia, complicated urinary tract infections and a variety of skin and tissue infections, in both healthy and immuno-compromised individuals ( Lee et al., 2017). Infections caused by multi-drug resistant (MDR) Acinetobacter species are among the most urgent threats to human health.
P.A. PBP3 INHIBITION DRIVERS
The following mini-review summarizes the molecular drivers of efficacy of this combination against this troublesome pathogen, with an emphasis on the biochemical features of each partner. This sulbactam-durlobactam combination is currently in late-stage development for the treatment of Acinectobacter infections, including those caused by carbapenem-resistant isolates, for which there is a high unmet medical need. However, when combined with durlobactam, the activity of sulbactam is effectively restored against these notoriously multidrug-resistant strains. Because sulbactam is also susceptible to cleavage by numerous β-lactamases, its clinical utility for the treatment of contemporary Acinetobacter infections is quite limited. The latter feature is due to sulbactam’s ability to inhibit certain penicillin-binding proteins, essential enzymes involved in bacterial cell wall synthesis in this pathogen. Sulbactam is a first generation β-lactamase inhibitor with activity limited to a subset of class A enzymes that also has direct-acting antibacterial activity against Acinetobacter spp.